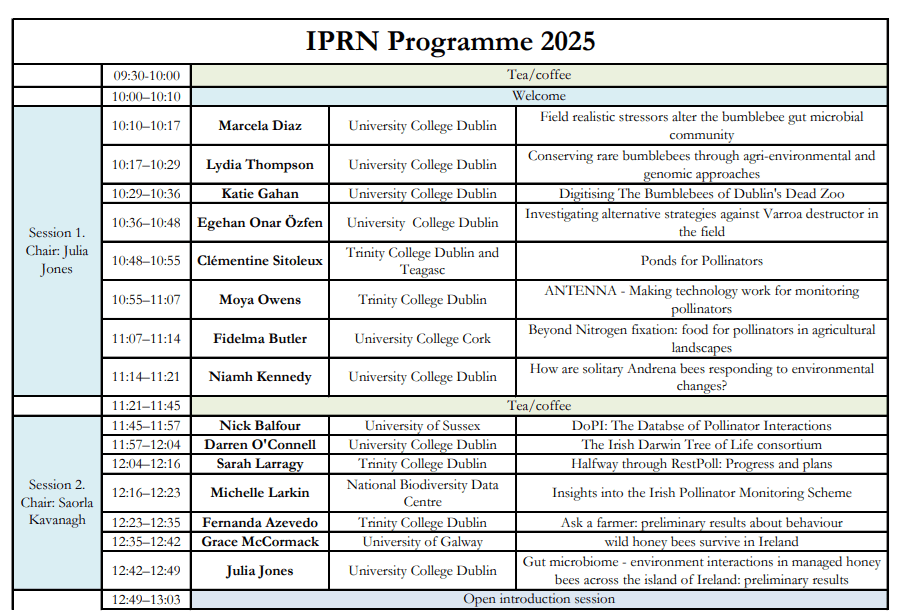
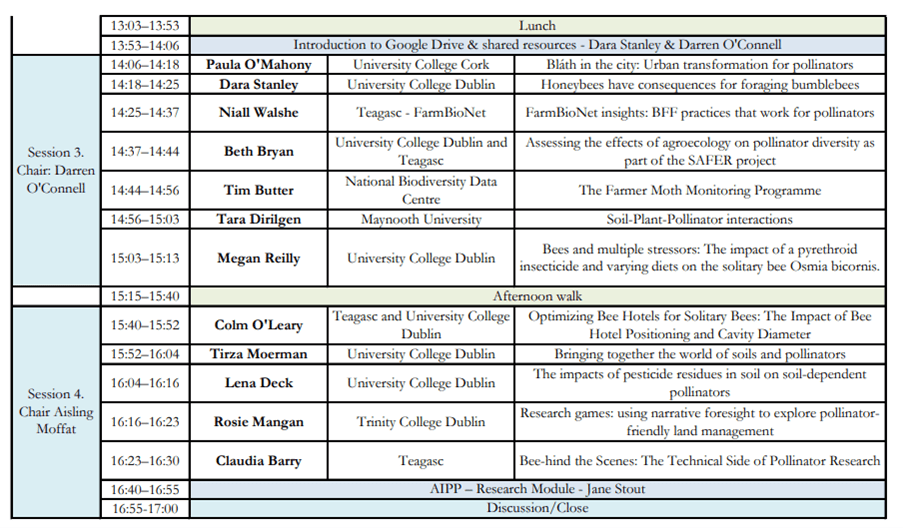
Written by PhD student Elaine Marshall, whose thesis focuses on gender in ecosystem restoration and management.

I recently returned from a 10 day trip to Ghana, participating in the ‘Birds Bees and Business’ project kick off, led by Vogelbescherming (VBN) ‘Birdlife Netherlands’, with Birdlife International in-country partners: Ghana Wildlife Society, the UK’s RSPB, Naturama (Burkina Faso), SOS Foret (Cote D’Ivoire), NABU (Birdlife Germany), as well as the ‘Fair Climate Fund’ (Netherlands) and ‘Tiigalpa’ (Burkina) NGOs, and a regionally important natural product social enterprise called ‘Vitara’.
Arriving in a hot, humid Accra, I met with the core team including project leader who I have worked with on and off since 2017. The following morning we flew Northwards to Tamale, and with the Harmattan winds blowing from the Sahara, the dust impeded much visibility both from the plane, and during the wider regional visit. Once all project partners had arrived in Tamale, and as the sun went down and the impossibly large fruit bats began their night patrols, we assembled in the evening cool to begin our journey working together, in French and English, introducing our technical backgrounds, organisations and interests. The next day we departed overland on a 3 hour drive to Mole National Park in Savannah Region, the location of our 6 day inception workshop.

Leaving the urban chaos of Tamale, you are immediately struck by the amount of degradation in the landscape, the absence of trees other than occasional small shea (Vitellaria paradoxa), baobab (Adansonia), African locust bean (Parkia biglobosa) or Kapok (Ceiba pentandra), the volumes of charcoal being sold on the side of the road (bulked up for collection by commercial scale intermediaries), grazing livestock – particularly goats, and areas of extensive ‘controlled’ burning.

The inception workshop was a carefully planned opportunity to enable us all to present our individual work and research, identify what we bring to the project including opportunities for synergy, support and partnership across the region, and input into a Theory of Change for the region, building on collective in-country partner progress, across the 3 project pillars of biodiversity conservation, socio economic well-being, and climate change adaptation and mitigation.

I presented a summary of my PhD research to date, including my conceptual framework drawing on feminist political ecology (FPE), outlined research questions and methodological approach for evaluating differentiated gender perspectives around restoration and sustainable land management (SLM). It generated lots of interest across the project partners, including interesting discussions around the sensitive nature of gender work, particularly in Islamic culture, and the importance of working with both women and men. We discussed the challenges of empowering women in ways which are culturally and socially acceptable, and how successful community engagement can enable sustained project outcomes.
We considered the possibilities of undertaking research in Cote D’Ivoire as well as Ghana, and also the relevance of previous work between 2016-2020 with Naturama colleagues in Burkina Faso. An opportunity to follow up and evaluate the perceived impacts by men and women, of farmer led interventions for nature restoration in Burkina Faso, would have been ideal. Sadly, regional insecurity and broken Diplomatic ties between Burkina and Europe, underpins Foreign Office advice not to travel, making this field work option challenging to insure. As such, for now the focus is on Ghana.
I set up meetings with the Ghana partners to discuss my community-based research in the Mole buffer zone. I wanted to assess the lie of the land, including ongoing and envisaged work, selection of research communities (location and number), and identify a research assistant, community ‘gatekeeper’, and access officer to help with translation. Physical infrastructure is limited in places to dirt roads, necessitating 4×4 vehicular access, and best avoided during the rainy season (June – September). We talked about the possibility of undertaking research in April, between Ramadan finishing and the rains beginning from mid May. Helpfully I was able to visit 5 communities in total and speak with some of the community women who are involved in shea production.
Communities in the Mole National Park (MNP) buffer zone have community resource management plans (CREMAS) which are developed and implemented with the support of Ghana Wildlife Society. Mole was established in 1956 and is the first and largest National Park in Ghana, covering 4577 km2, located in the Guinea savanna vegetation zone. A variety of mammals, including elephants, birds and tree species, attract international tourists. A history of conflict exists between local communities and MNP, tracing back to enforced eviction and involuntary relocation, at the time of protected area gazetting. The establishment of MNP resulted in the loss of hunting grounds, farmland, and sacred sites, with notable impact on women through reduced access to traditional harvesting sites for important non timber forest products (NTFPs), including shea. Cultural norms which alienate women from accessing land and making decisions relating to natural resources, reinforce inequalities and exacerbate the impact of protected area conflicts on women: “Land issues are men’s issues—we women have little say in matters of land” (Soliku, 2024). Wildlife laws prohibiting local people from hunting and accessing other resources in the park, as well as ongoing human wildlife conflict around elephants and crops, further aggravates the relationships between local communities, chiefs, park and government officials. Households in the region are predominantly male headed (90%), with sociocultural norms determining women’s access to productive resources, participation in decision-making, and expected caregiving roles (Nyarko, 2002). Male economic migration to the South of Ghana continues, leaving behind wives (and children) who assume dual roles as caregivers and breadwinners.
I spoke with community women and men in all 5 communities, around shea production and the importance of the landscape in meeting wider livelihood needs, from fuelwood and water, to food and income. Women consistently reported unreliable rains resulting in drought last year and lost harvests. The importance of shea in terms of helping with ‘income smoothing’ for households is significant, as were observations that the availability and abundance of nuts is declining. “ Before, ten years back, we used to collect all that we needed and there were still so many nuts left on the ground. Now we struggle to find what we need….” Local women attributed this shortage to unpredictable rains and a changing climate, but previous research in neighbouring Burkina confirming that shea is insect pollinated, also identified pollination limitation across shea sample plots, and this was greatest where plant diversity was lowest and least able to support healthy pollinator populations (Delaney et al., 2020).

Reflecting on methodological approach and research questions:
- The wider project partnership recognises the importance of working with men and women, sharing valuable insight around the importance of respect for cultural practices in interventions which seek to empower women. Many ongoing projects have elements of gender mainstreaming incorporated.
- There are partner limitations in capacity and personnel, but I met with 2 excellent local staff whom I hope to engage full time to work with during the next field work visit.
- I will use the FPE (feminist political ecology) framework to provide an approach for evaluating gendered perceptions of landscape management, risk, resilience, benefit sharing, decision-making, and changes over time, etc.. The scoping visit has been enlightening as to what elements of different lived experiences the case study communities are best able to talk about, and what areas might provide most insight around gendered community dynamics.
- Interventions to increase food security through vegetable gardens and bee keeping are underway, and restoration activities are being established in the communities, with community tree nurseries leading the way following consultation around species selection.
- The data collection will be a mixed methods approach collecting both qualitative and quantitative data using: surveys, semi structured interviews with key informants; focal groups with both men and women; my observations and journal notes; and time use maps. All communities have women who collect, process and sell shea (as members of a producer group), and these women will provide my targeted sample across 3 different communities, Male key informants will be identified from each community, and additional perspectives gleaned from male and female focus groups.
- Deliberately parking the language of ‘gender equality’, to enable a more nuanced exploration of ‘gendered perceptions around wider community resilience and livelihood strategies’, when working in the field.
Using a gender lens to identify and explore how people interact with their natural environment:
- Gender roles in income generation, farming and food production, and natural resource management? What are the gendered priorities, opportunities, challenges?
- What does a typical day look like? Who does what, where, and why?
- What decisions are made and how and who makes them? And how has all of this changed over time? Observing any areas of tension or conflict.
- What does a typical day look like? Who does what, where, and why?
Thematic areas for discussion include: access to resources and land; restoration and sustainable land management; labour and care burdens; livelihoods and income (agricultural, shea, other NTFPs etc).
And finally, we were lucky and enjoyed a couple of safaris around MNP near to our lodge, early morning and evening, and a special opportunity to walk to the watering hole. We saw elephants, wart hogs, crocodiles, antelopes, monkeys, and huge bird diversity, from fly catchers, to kingfishers, to martial eagles and vultures, and on an evening drive, some very cute bush babies!

Delaney, A., Dembele, A., Nombré, I., Gnane Lirasse, F., Marshall, E., Nana, A., Vickery, J., Tayleur, C., Stout, J.C., 2020. Local‐scale tree and shrub diversity improves pollination services to shea trees in tropical West African parklands. Journal of Applied Ecology 57, 1504–1513. https://doi.org/10.1111/1365-2664.13640
Nyarko, P., 2002. Navrongo DSS, Ghana,. Population and Health in Developing Countries in vol. 1, Population, Health, and Survival at INDEPTH Sites 247–256. https://www.indepth-network.org/Resource%20Kit/INDEPTH%20DSS%20Resource%20Kit/LinkedDocuments/INDEPTH%20Monograph%20I%20Ch1-7%20Introduction,%20Methods%20&%20Life%20Tables.pdf.
Soliku, O., 2024. Living around Mole National Park: Grassroots Perspectives on Gendered Experiences of Protected Area Conflicts and Peacebuilding Strategies.